Atomic Properties and Periodic Trends
Figure 1

Figure 2

Figure 3
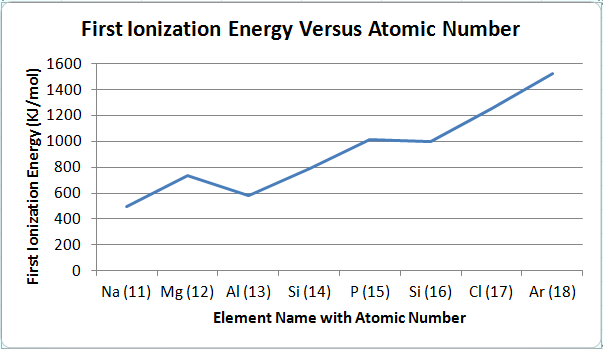
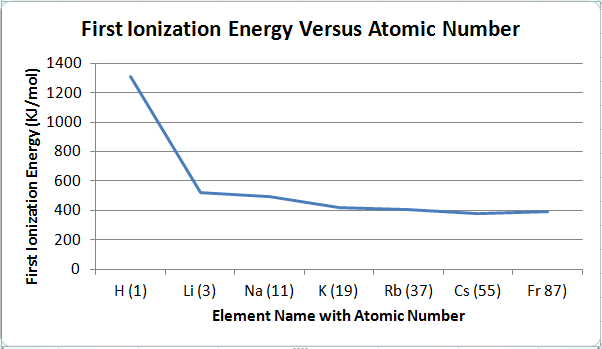
Figure 4
1. Atomic Radius - the average distance from the atom's nucleus to the outermost boundary of the electron cloud; the measurement of size of an atom
2. The atomic radius decreases when going from left to right on the periodic table and increases when going from top to bottom on the periodic table. Therefore, the element with the largest atomic radius would be located on the most bottom-left space, and the element with the smallest atomic radius would be located on the most top-right space. The evidence that the atomic radius decreases when going from left to right (also increasing when going from right to left) on the periodic table is seen in Figure 1. Figure 1 shows the relationship between the atomic radius and the elements in row three. The elements are in order by increasing atomic number: Na (11) Mg (12)
Al (13)
Si (14)
P (15)
Si (16)
Cl (17)
Ar (18). The atomic radius for Na is 180 pm, and the atomic radius for Ar is 71 pm. The remaining elements' atomic radii are decreasing from the atomic radius of Na to the atomic radius of Ar. As the atomic number increases in a row, the atomic radius decreases. The evidence that the atomic radius increases when going from top to bottom on the periodic table is seen in Figure 2. Figure 2 shows the relationship between the atomic radius and the elements in group one. The elements are in order by increasing atomic number: H (1)
Li (3)
Na (11)
K (19)
Rb (37)
Cs (55)
Fr 87). The atomic radius for H is 25 pm, and the atomic radius for Fr is 270 pm. The remaining elements' atomic radii are increasing from the atomic radius of H to the atomic radius of Fr. As the atomic number increases in a family, the atomic radius increases.
3. There are many factors that determine these trends in atomic radius. The atomic radius increases when going from top to bottom on the periodic table because the number of energy levels increases. Since each energy level is further than the last, it results in the electrons being further from the nucleus, causing a larger atomic radius. The atomic radius decreases when going from left to right on the periodic table because as one goes across the periodic table, in addition to one electron being added, one proton is also added. The addition of each proton results in a greater force of attraction, pulling in the electrons closer.
4. The trend in ionic radii of period three and group one would be similar to the results observed for atomic radii, assuming that all of the elements become anions or that all of the elements become cations. Anions are the addition of electrons to an element. The trend in the graphs would be similar; the only difference would be that the atomic radius would be larger. The nucleus would not be able to pull in the electrons as tightly as before, and there is greater electron-electron repulsion due to the addition of electrons, resulting in a larger atomic radius. Cations are the subtraction of electrons to an element. The trend in the graphs would be similar; the only difference would be that the atomic radius would be smaller. The nucleus would be able to pull in the electrons tighter than before because there are less electrons, and there is less electron-electron repulsion, which results in a smaller atomic radius. Also, if an element becomes a cation that has the same number of electrons as a noble gas, the atomic radius is significantly smaller since it lost an entire energy level.
5. First Ionization Energy - the energy required to remove the outermost (highest energy) electron
6. The first ionization energy increases when going from left to right on the periodic table and decreases when going from top to bottom on the periodic table. Therefore, the element with the largest first ionization energy would be located on the most top-right space, and the element with the smallest first ionization energy would be located on the most bottom-left space. The evidence that the first ionization energy increases when going from left to right (also decreasing when going from right to left) on the periodic table is seen in Figure 3. Figure 3 shows the relationship between the first ionization energy and the elements in row three. The elements are in order by increasing atomic number: Na (11) Mg (12) Al (13) Si (14) P (15) Si (16) Cl (17) Ar (18). The first ionization energy for Na is 496 KJ/mol, and the first ionization energy for Ar is 1521 KJ/mol. The remaining elements' first ionization energies are increasing from the first ionization energy of Na to the first ionization energy of Ar. As the atomic number increases in a row, the first ionization energy increases as well. The evidence that the first ionization energy decreases when going from top to bottom on the periodic table is seen in Figure 4. Figure 4 shows the relationship between the first ionization energy and the elements in group one. The elements are in order by increasing atomic number: H (1) Li (3) Na (11) K (19) Rb (37) Cs (55) Fr 87). The first ionization energy for H is 1312 KJ/mol, and the first ionization energy for Fr is 393 KJ/mol. The remaining elements' first ionization energies are decreasing from the first ionization energy of H to the first ionization energy of Fr. As the atomic number increases in a group, the first ionization energy decreases.
7. There are many factors that determine these trends in first ionization energy. The first ionization energy decreases when going from top to bottom on the periodic table because of the "shielding" effect, the electrons in lower energy levels blocking the force of attraction by the protons on the electrons in the most outer energy level. These electrons in the lower energy levels essentially act as a "shield," hence the term. Since the electrons furthest from the nucleus are the easiest to remove, the first ionization energy decreases when going from top to bottom. The first ionization energy increases when going from left to right on the periodic table because as more protons are added, the force of attraction upon the electrons are greater, resulting in a smaller atomic radius. The electrons furthest from the nucleus are greatly attracted to the nucleus, making it harder to remove the outermost electron, which causes the first ionization energy to increase when going from left to right. There are line in Figure 3 is not smooth due to orbitals. The first break is between Mg and Al. This is because it is easier to remove the lone electron in the p orbital rather than to remove an electron in the filled s orbital. The second break is between P and Si. This is because once the p orbital has been half filled, the next electron must pair up with one of the other electrons in the p orbital. Due to electron-electron repulsion, energy to remove this electron is less.
8. The trend in electron affinities of the elements in period three and group one would are similar to the results I observed for ionization energies. The electron affinity increases when going from left to right on the periodic table and decreases when going from top to bottom on the periodic table. Electron affinity is the energy change associated with the addition of an electron. The electron affinity decreases when going from top to bottom on the period table because of the addition of energy levels. There is a weaker attraction between the nucleus and the electrons on the furthest energy level. Therefore, it takes less energy to add that electron to another atom. The electron affinity increases when going from left to right on the periodic table because atoms want to have eight valence electrons on their energy levels, causing a greater release of energy as they get closer and closer to a filled valence shell (eight electrons). Therefore, the electron affinity increases when going from left to right. There are also breaks in the line when the elements in the same row are graphed versus the electron affinity. The breaks occur right after the s orbital is filled, the p orbital is half full, and at the noble gases. It actually requires energy in order for the atoms to gain electrons in these cases.
9. Metals - At room temperature, metals are a shiny solid. The exception is mercury, which is a shiny liquid at room temperature. They have high melting points and high densities. They can be deformed without breaking. Most metals are malleable and ductile. Most metals have a large atomic radius, low ionization energy, and low electronegativity. Their valence electrons can be removed fairly easily.
Nonmetals - Nonmetals are generally poor conductors of electricity and heat. Most nonmetals gain electrons easily. They have high ionization energies and high electronegativities. Solid nonmetals are not shiny and are brittle. They usually have a small atomic radius.
Metalloids - The electronegativity, ionization energy, and atomic radius are between metals and nonmetals. Metalloids have characteristics of both metals and nonmetals. Their densities, melting points, and boiling points vary. They make good semiconductors. Their reactivity depends on what they are reacting with.
Noble gases - Noble gases are not reactive due to their completely filled valence shell. They have high ionization energies and barely have any electronegativity. They tend to not gain or lose electrons. They are all gases at room temperature and have low boiling points.
Nonmetals - Nonmetals are generally poor conductors of electricity and heat. Most nonmetals gain electrons easily. They have high ionization energies and high electronegativities. Solid nonmetals are not shiny and are brittle. They usually have a small atomic radius.
Metalloids - The electronegativity, ionization energy, and atomic radius are between metals and nonmetals. Metalloids have characteristics of both metals and nonmetals. Their densities, melting points, and boiling points vary. They make good semiconductors. Their reactivity depends on what they are reacting with.
Noble gases - Noble gases are not reactive due to their completely filled valence shell. They have high ionization energies and barely have any electronegativity. They tend to not gain or lose electrons. They are all gases at room temperature and have low boiling points.
10. Metals - Na, Mg, Al
Metalloids - Si
Nonmetals - P, S, Cl, Ar
Noble Gases - Ar
Metalloids - Si
Nonmetals - P, S, Cl, Ar
Noble Gases - Ar
11. All atoms aim to have eight valence electrons. Group one elements have only one electron on its most outer shell. They need to lose that one electron in order to reach eight outer valence electrons and to become stable. Because they desire getting rid of that electron, they are the most active metals, trying to give it away to any element it meets. Also, as one goes down the group one family, the atomic radius increases due to an addition of an energy level. This means that the electron is further from the nucleus, receiving a weaker force attraction by the protons. Therefore, it is even easier for group one metals with higher energy levels to lose an electron to have eight valence electrons on its most outer shell, making them the most active metals.
Group seventeen is the most active nonmetals because just like the group one metals, group seventeen nonmetals are trying to achieve eight valence electrons on its outermost energy level. It has seven valence electrons already, and it needs eight to be stable; therefore, it needs to take one electron from another atom, resulting in it being the most active nonmetals.
Group seventeen is the most active nonmetals because just like the group one metals, group seventeen nonmetals are trying to achieve eight valence electrons on its outermost energy level. It has seven valence electrons already, and it needs eight to be stable; therefore, it needs to take one electron from another atom, resulting in it being the most active nonmetals.
1.
2. Based off of my periodic table that I created, an element with an atomic mass of 11.29 that has nonmetallic properties and is very reactive would have an atomic number of 5.
3. Based off of my periodic table that I created, an element having an atomic mass of 15.02 that exhibits metallic properties but is softer than lambda and harder than beta would have an atomic number of 8.
4. The elements towards the left of the table are metal. The elements towards the right of the table are nonmetals. Excluding the column that Gamma is in, the columns on the ends of the table contain the elements that are the most reactive. In the metal side of the table, as the elements get closer to the left most column, they become harder. The Epsilon family is very reactive, as well as the Beta family. The atomic mass and atomic number increases as one goes from the left side of the periodic table to the right side of the periodic table. It is understood that as one goes from left to right on the periodic table, the atomic radius decreases, and as one goes from top to bottom on the periodic table, the atomic radius increases. This, in turn, means that as one goes from left to right on the periodic table, the first ionization energy and electronegativity increases, and as one goes from top to bottom on the periodic table, the first ionization energy and electronegativity decreases.
5. Beta would have the largest atomic radius. Delta would have the smallest atomic radius. Delta would have the highest first ionization energy. Beta would have the lowest ionization energy.

