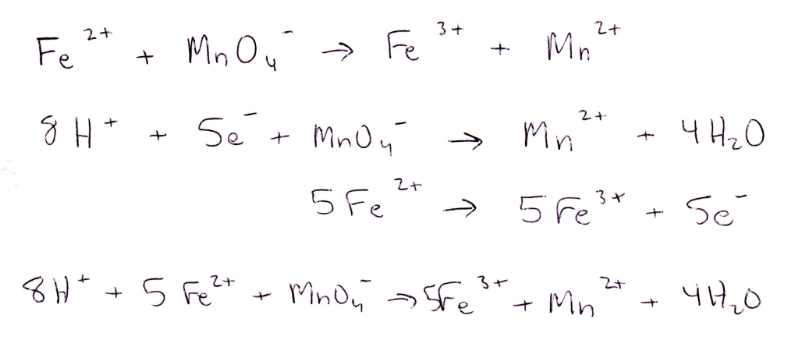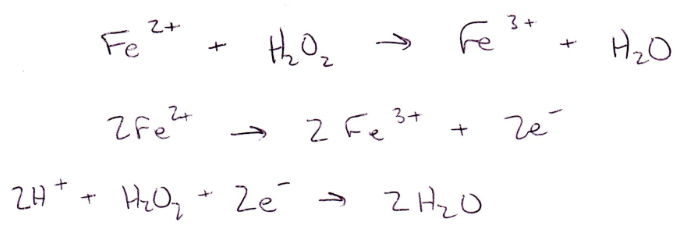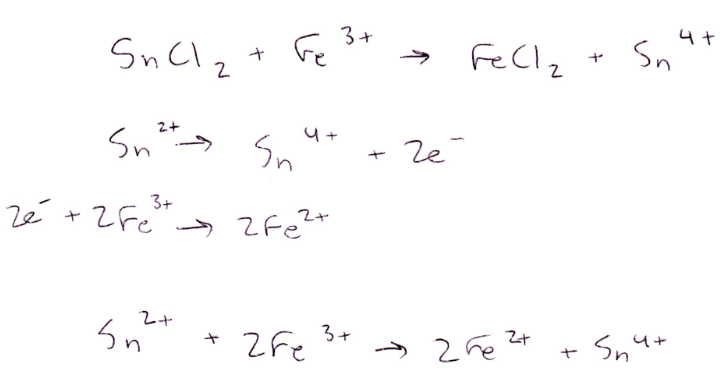Oxidation-Reduction Reactions Lab
Purpose: to run three experiments where a compound which is colorless in solution when reduced is converted to a deeply colored solution when oxydized in order to achieve a more in-depth understanding of oxidation-reduction reactions.
Data Table
PartPart 1
Part 2
|
ObservationsClear > Amber > Black > Dark Violet
Clear > Dark Blue > Clear > Dark Blue
|
Conclusion
At first, both the beaker with the KMnO4 solution and the beaker with the H2O2 solution appeared to be clear. Then, after the 10 mL of the potassium thiocyanate solution was added to each beaker, both solutions appeared to be a transparent amber color. After the potassium permanagate solution was added to the beaker labeled KMnO4, the solution turned black. The same goes for the beaker labeled H2O2 after the hydrogen peroxide solution was added. Adding stannous chloride solution to each beaker while mixing made the solutions in each beaker turn dark violet. When the potassium hydroxide solution was diluted with de-ionized water, the solution was clear. After the glucose was added, the solution still remained clear. After the methylene blue solution was mixed in, the solution became dark blue. After 10 minutes, the solution became clear. After mixing the solution again, the solution became dark blue, and after 10 minutes of allowing the flask to be still, the solution became clear.
Discussion of Theory
Oxidation is the loss of elections, and reduction is the gain of electrons. Oxidation and reduction reactions can be compared to acid-base reactions because acid-base reactions always occur in pairs. The H+ cation of an acid reacts with the OH- anion of a base. Electrons do not disappear; an electron must be gained by another molecule for every electron lost by a molecule in a reaction. These reactions occur as a set, so oxidation and reduction reactions and called redox reactions for short. Redox reactions are essentially two half-reactions, an oxidation half and a reduction half. The oxidation half loses electrons, and the reduction half gains electrons. Batteries and power cells use redox reactions. Sometimes, the effects of redox reactions can be observed physically. Depending whether they are in an oxidized or reduced state, their properties vary. In the presence of thiocyanate, the oxidation of a Fe2+ containing compound to Fe3+ will form a red color. Oxidation of the dye methylene blue will form a deep blue color. Methylene blue is a leuco dye, which means that its molecules can exist in two forms, one form is colored and the other form is colorless.
The number of electrons gained is equal to the number of electrons lost in redox reactions. Some examples of redox reactions include the oxidation of fats, sugars, and proteins for energy. For a free element, the oxidation number of the atom is 0. For a monatomic ion, the oxidation number equals its charge. Oxygen has an oxidation number of -2 in compounds except in hydrogen peroxide, where the oxidation number of oxygen is -1. Hydrogen has an oxidation number of +1 in compounds containing hydrogen. Fluorine has an oxidation number of -1 in compounds. For an electrically neutral compound, the sum of the oxidation states must be equal to zero. The oxidized substance is the reducing agent, and the reduced substance is the oxidizing agent. Metals are reducing agents. Potassium is the best reducing agent and worst oxidizing agent among metals.
In Part 1, Fe2+ is oxidized into Fe3+ after reacting with permanganate and hydrogen peroxide. Fe2+ is oxidized into Fe3+ when reacting with permanganate because Mn7+ to Mn2+ has a higher reduction potential than Fe2+ to Fe3+. Fe2+ is oxidized into Fe3+ when reacting with hydrogen peroxide because O1- to O2- has a higher reduction potential than Fe2+ to Fe3+. When reacted with stannous chloride, Fe2+ is oxidized into Fe3+ because Sn2+ to Sn4+ has a higher reduction potential than Fe2+ to Fe3+.
The number of electrons gained is equal to the number of electrons lost in redox reactions. Some examples of redox reactions include the oxidation of fats, sugars, and proteins for energy. For a free element, the oxidation number of the atom is 0. For a monatomic ion, the oxidation number equals its charge. Oxygen has an oxidation number of -2 in compounds except in hydrogen peroxide, where the oxidation number of oxygen is -1. Hydrogen has an oxidation number of +1 in compounds containing hydrogen. Fluorine has an oxidation number of -1 in compounds. For an electrically neutral compound, the sum of the oxidation states must be equal to zero. The oxidized substance is the reducing agent, and the reduced substance is the oxidizing agent. Metals are reducing agents. Potassium is the best reducing agent and worst oxidizing agent among metals.
In Part 1, Fe2+ is oxidized into Fe3+ after reacting with permanganate and hydrogen peroxide. Fe2+ is oxidized into Fe3+ when reacting with permanganate because Mn7+ to Mn2+ has a higher reduction potential than Fe2+ to Fe3+. Fe2+ is oxidized into Fe3+ when reacting with hydrogen peroxide because O1- to O2- has a higher reduction potential than Fe2+ to Fe3+. When reacted with stannous chloride, Fe2+ is oxidized into Fe3+ because Sn2+ to Sn4+ has a higher reduction potential than Fe2+ to Fe3+.
Data Analysis Questions
1.
The reduction potential of MnO4- at 25 oC for MnO4- + 8H+ + 5e- → Mn2+ + 4H2O is 1.51V, and the reduction potential of H2O2 at 25 oC for H2O2 + 2H+ + 2e- → 2H2O is 1.77V. MnO4⁻ has a lower reduction potential than H2O2 so it is less likely to be reduced than H2O2.
2.
The Cl2 is a spectator ion, so it is not included in the balanced ionic equation.
4.
In the solution containing potassium hydroxide, de-ionized water, and glucose, glucose and OH- react to form glucoside, a powerful reducing agent, which causes methylene blue to change from the colored form to the colorless form. When the flask is shaken, oxygen gas is introduced to the solution. Oxygen is reacted with the colorless solution, which causes the methylene blue to return to its colored form.
5.
Glucose and OH- react to form glucoside, a powerful reducing agent, which causes methylene blue to change from the colored form to the colorless form. When oxygen is introduced to the solution, methylene blue returns to its colored form. After 10 cycles, the color remains clear because there are no more electrons that can be transferred. There is an unlimited supply of oxygen gas which becomes O2- but there is a limited supply of electrons in the solution, and there comes to be a point where no more electrons can be transferred, so the solution remains colorless.