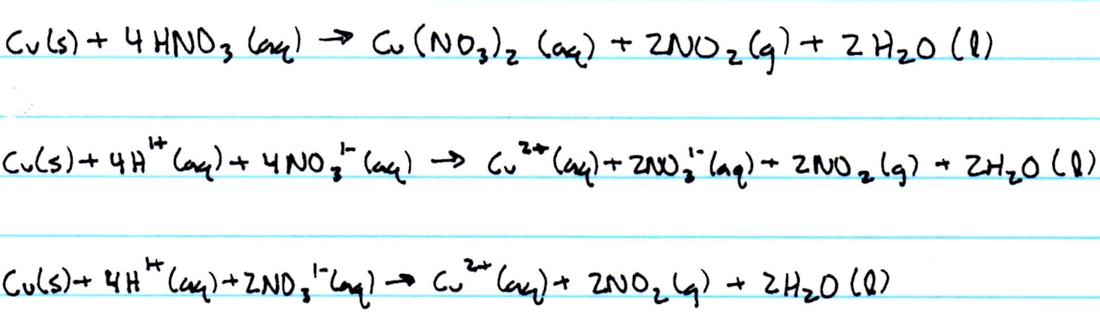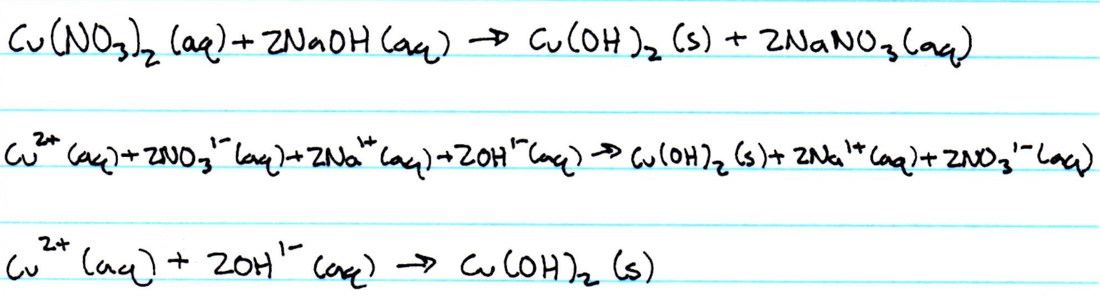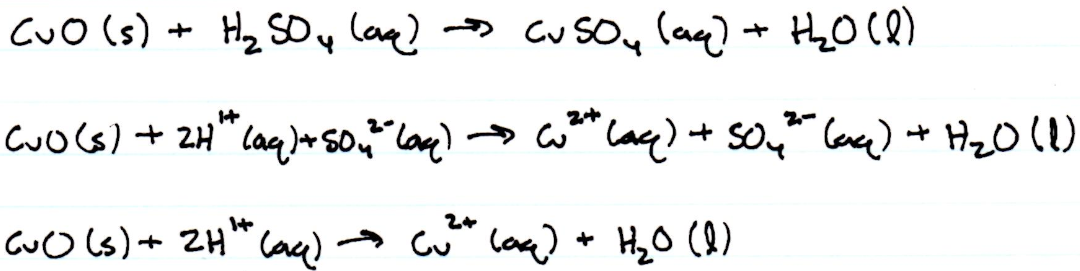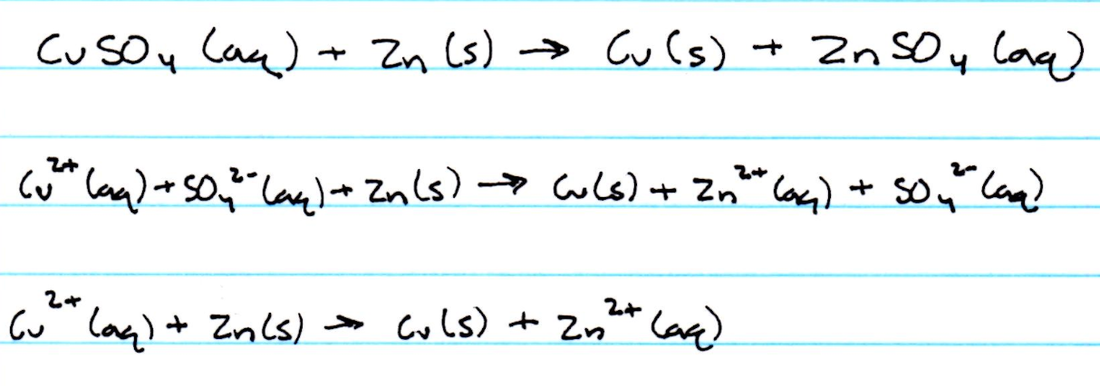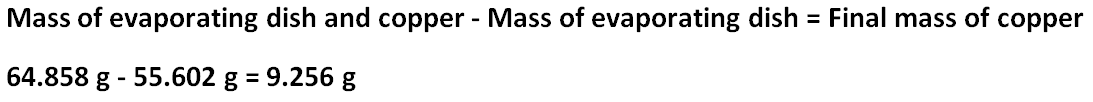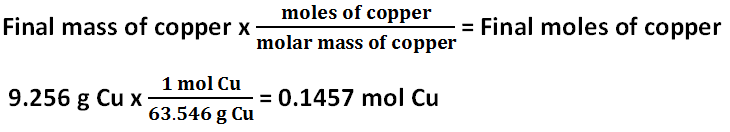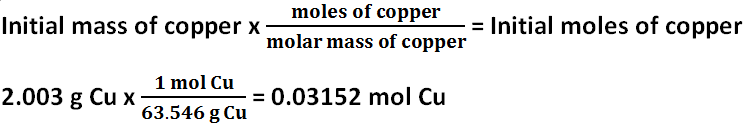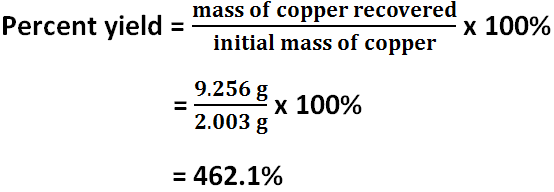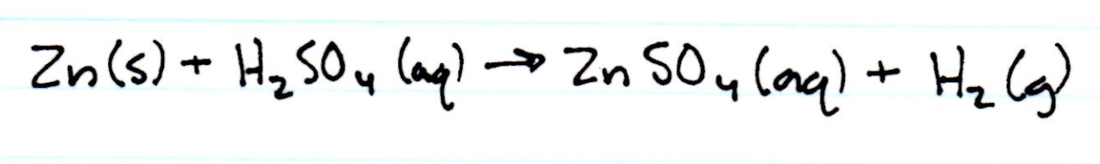Stoichiometry Using Copper Lab
Purpose
To demonstrate the conservation of mass involving chemical reactions using copper and its compounds
Qualitative and Quantitative Data
Quantitative data
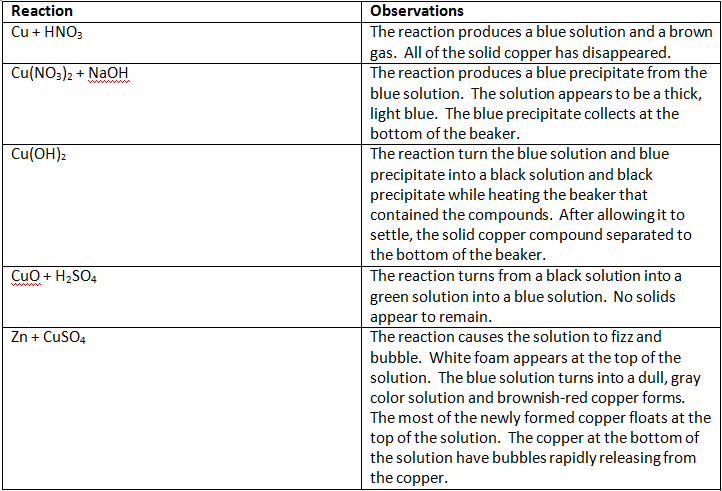
Qualitative data
Net Ionic Equations
Calculations
Final mass of copper
Final moles of copper
Initial moles of copper
Percent yield
Conclusions
The initial mass of copper was 2.003 grams. The final mass of copper was 9.256 grams of copper. The initial and final masses of copper are supposed to be the same, but they are different. The initial moles of copper is 0.03152 mol, and the final moles of copper is 0.1457 mol. The initial and final moles of copper are supposed to be the same, but they are different. There must have been error because both the initial moles and masses of copper and the final moles and masses of copper should have been very similar, if not exactly the same, in their results, but the moles and masses of copper vary significantly. The percent yield of copper is 462.1%.
Discussion of Theory
This experiment was to prove the law of conservation of mass. The initial mass of copper was supposed to be exactly the same as the final mass of copper. However, in this experiment, the results were completely different due to human error and other sources of error listed below. Theoretically, the masses of cooper were supposed to be identical. Since the masses were supposed to be the same, that means that the initial moles of copper and the final moles of copper were supposed to be identical also because mass can be converted into moles.
This experiment also proved the solubility rules. All of the nitrate compounds, such as copper nitrate and sodium nitrate, were aqueous, and all of the sulfate compounds, such as copper sulfate and zinc sulfate, were aqueous. The hydroxide compounds were aqueous only sometimes, in the case of it being bonded to a group I metal. In other cases, such as when the hydroxide was bonded with copper, it was a solid - insoluble.
The ions of elements created unique colors in the solution. Some ions do not produce a color in a solution, remaining colorless; however, some ions do change the color of the solution. In this experiment, the copper ions produced a light blue/green color. The characteristic of a copper ion is that it turns solutions into a blue/green color. This characteristic is useful in determining what a blue/green solution may contain. Other ions produce colorful solutions, and they can be used to predict what may be in other solutions as well.
There were many different types of reactions involved in this experiment. When copper reacts with a strong base, in this case NaOH, the reaction forms metal hydroxides, and these reactions are replacement reactions. Metal hydroxides are mostly insoluble. Copper hydroxide forms a blue jelly substance. Decomposition is when a compound is broken into smaller pieces, such as when CuO decomposed. There is evidence that a reaction is occurring. In this experiment, it varied from the formation of a gas, the formation of a precipitate, and the change of color only to name a couple of physical changes.
This experiment also proved the solubility rules. All of the nitrate compounds, such as copper nitrate and sodium nitrate, were aqueous, and all of the sulfate compounds, such as copper sulfate and zinc sulfate, were aqueous. The hydroxide compounds were aqueous only sometimes, in the case of it being bonded to a group I metal. In other cases, such as when the hydroxide was bonded with copper, it was a solid - insoluble.
The ions of elements created unique colors in the solution. Some ions do not produce a color in a solution, remaining colorless; however, some ions do change the color of the solution. In this experiment, the copper ions produced a light blue/green color. The characteristic of a copper ion is that it turns solutions into a blue/green color. This characteristic is useful in determining what a blue/green solution may contain. Other ions produce colorful solutions, and they can be used to predict what may be in other solutions as well.
There were many different types of reactions involved in this experiment. When copper reacts with a strong base, in this case NaOH, the reaction forms metal hydroxides, and these reactions are replacement reactions. Metal hydroxides are mostly insoluble. Copper hydroxide forms a blue jelly substance. Decomposition is when a compound is broken into smaller pieces, such as when CuO decomposed. There is evidence that a reaction is occurring. In this experiment, it varied from the formation of a gas, the formation of a precipitate, and the change of color only to name a couple of physical changes.
Sources of Error
The yield of copper for the experiment was too high due to many sources of error. The copper was originally placed into a 100 mL beaker rather than a 250 mL beaker; therefore, after the sodium hydroxide was added to the solution, the new solution was transferred to the 250 mL beaker, resulting in small fragments unable to be transferred to the 250 mL beaker. This should have lowered the final mass of copper because the small fragments were a copper compound. When heating the beaker, the solution in the beaker began to boil and burst its bubbles. The bubbles popped, some of it leaving the beaker unto the surrounding area. This should have lowered the final mass of copper because the solution that the membrane of the bubble consisted of a copper compound. While decanting, some members of the group could not differentiate the copper in the solution, mainly because solution was black and its components were not visible. Therefore, about a quarter of the copper compound left the beaker while decanting. This should have lowered the final mass of copper because almost a quarter of the whole copper compound was decanted along with the water. When sulfuric acid was added, not all of the copper compound was affected by it because some of it was still left on the sides of the beaker. Therefore, when zinc was added to the new solution, not all of the copper was involved in the reaction and not all of the potential copper was produced. This should have lowered the final mass of copper because the copper produced was not produced using all the copper compound. Also, when the copper was weighed to determine its final mass, there was a gelatin-like substance on the edges of the newly formed copper on the evaporating dish. This should have raised the the measured final mass of copper because it was not supposed to be there, but it was included in the measurement anyway. And it did have a profound affect on the measured final mass of copper, increasing it significantly.
Questions
1. The product of the reaction between copper and nitric acid in step 2 is placed on ice in order to cool the solution. Only after cooling the solution did the solid product appear in the solution. Therefore, the product is placed in ice to aid in the quickness of the formation of the solid product, copper hydroxide.
2. The reaction that occurred in step 4 was a double replacement reaction. The reaction that occurred in step 7 was a dehydration reaction. The reaction that occurred in step 9 was a single replacement reaction.
3. The excess zinc is reacted with sulfuric acid to ensure that none of the excess zinc, which is a solid, remains a solid and that it is turned into zinc sulfate, which is an aqueous solution that can be poured out with water. If this was an incomplete reaction, there would be solid zinc still remaining. It would be weighed along with the copper precipitate, and the solid zinc would significantly increase the measured final mass of the copper, resulting in a larger mass than the initial mass of copper.
2. The reaction that occurred in step 4 was a double replacement reaction. The reaction that occurred in step 7 was a dehydration reaction. The reaction that occurred in step 9 was a single replacement reaction.
3. The excess zinc is reacted with sulfuric acid to ensure that none of the excess zinc, which is a solid, remains a solid and that it is turned into zinc sulfate, which is an aqueous solution that can be poured out with water. If this was an incomplete reaction, there would be solid zinc still remaining. It would be weighed along with the copper precipitate, and the solid zinc would significantly increase the measured final mass of the copper, resulting in a larger mass than the initial mass of copper.
4. When the CuO was washed, the ions that were removed were the excess hydroxide ions that could have remained after all of the copper reacted from the previous reaction.
5. After the sulfuric acid was added to the beaker, the form of copper that resulted was copper ions with a 2+ charge. Initially, before the reaction, the copper was in the copper compound, copper (II) oxide.
6. When the precipitated copper was washed, zinc ions were removed. The reaction involved the reactants copper sulfate and zinc. Copper sulfate was aqueous and zinc was solid. After the reaction occurred, the products were copper and zinc sulfate. Copper was solid and zinc sulfate was aqueous. After finding the net ionic equation, the products were solid copper and aqueous zinc ions. Therefore, when the copper was washed, the zinc ions were washed out with the water.
5. After the sulfuric acid was added to the beaker, the form of copper that resulted was copper ions with a 2+ charge. Initially, before the reaction, the copper was in the copper compound, copper (II) oxide.
6. When the precipitated copper was washed, zinc ions were removed. The reaction involved the reactants copper sulfate and zinc. Copper sulfate was aqueous and zinc was solid. After the reaction occurred, the products were copper and zinc sulfate. Copper was solid and zinc sulfate was aqueous. After finding the net ionic equation, the products were solid copper and aqueous zinc ions. Therefore, when the copper was washed, the zinc ions were washed out with the water.